This is a busy time of year for vernal pools! Amphibians often take the spotlight when talking about species that depend on these special habitats, but there’s much more going on under the surface! Educator Bridget Bradshaw made this handy ID guide to things you might see in a vernal pool near you.
Our Young Scientists Research Experience is one of our favorite programs of the year! Middle and high school students spend a week with us doing their own independent research projects. They come up with a research question, collect data to answer their questions, and share their findings during a presentation to friends and family.
We’re incredibly proud of the work these students did, and we’re excited to share their results with you!
If you’d like to sign up for next year’s Young Scientist Research Experience, you can subscribe to our email newsletter to be notified when registration opens.
Here’s a quick summary of all their hard work:
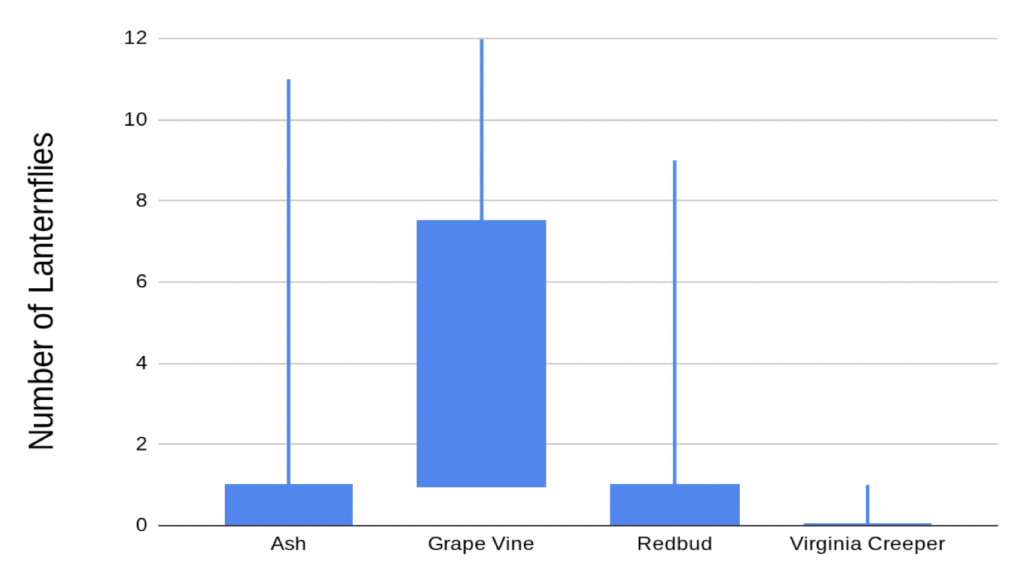
After noticing the many Spotted Lanternflies at Clifton this summer, Alistair, a second time Young Scientist, wondered if there are certain types of plants they prefer over others. Since Spotted Lanternflies are known to use Tree of Heaven and Tree of Heaven has compound leaves, Alistair decided to compare plants with compound and simple leaves to see if the insects might be seeking out leaves similar to their host plants and to compare vines and trees to see if the insects could tell what kind of plant they were landing on. While he didn’t find a big difference between plants with compound and simple leaves, he did find that there were lots of lanternflies on grape vines. Alistair also tried to lure in lanternflies with fake leaves of different colors and textures, but only two lanternflies were fooled. Although the experiment didn’t work, it’s always worth trying a little crafting in the name of science!

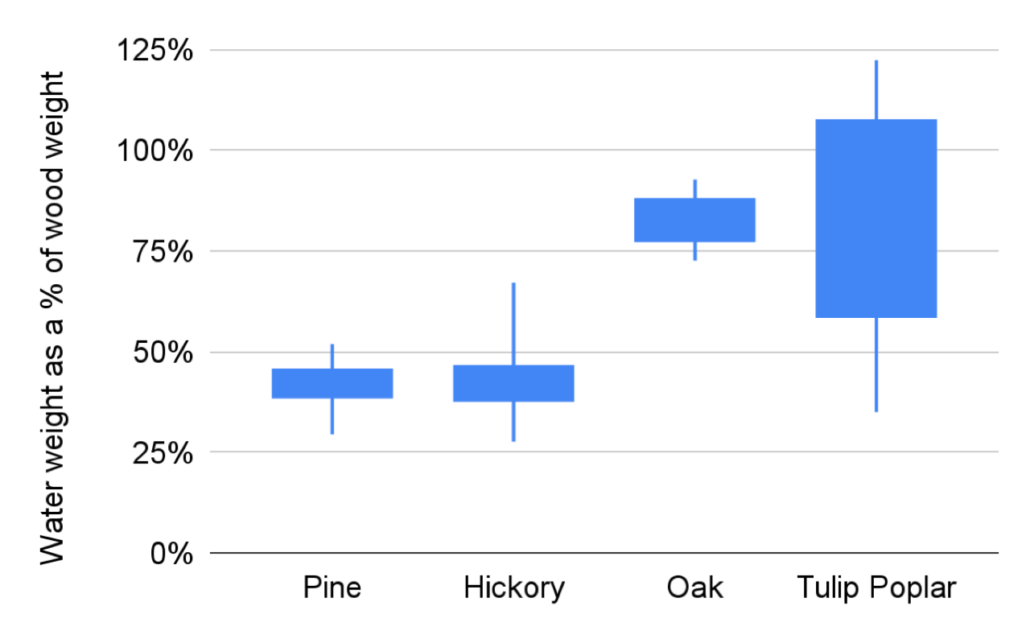
Lane, a fourth time Young Scientist and plant enthusiast, got excited about tree sap after noticing how wet a wood core was on our exploratory hike. He decided to see if different trees had different amount of saps (by measuring how wet their cores were), if different tree saps had different pHs, and if either factor was affected by the soil. Lane found that Tulip Poplars had the most sap by weight and pine trees had the least. Surprisingly, neither the moisture of the wood nor its pH was affected by where the trees were growing. Looking at Lane’s data, what we want to know next is if trees that grow quickly, like Tulip Poplars, tend to have more sap to facilitate that rapid growth!

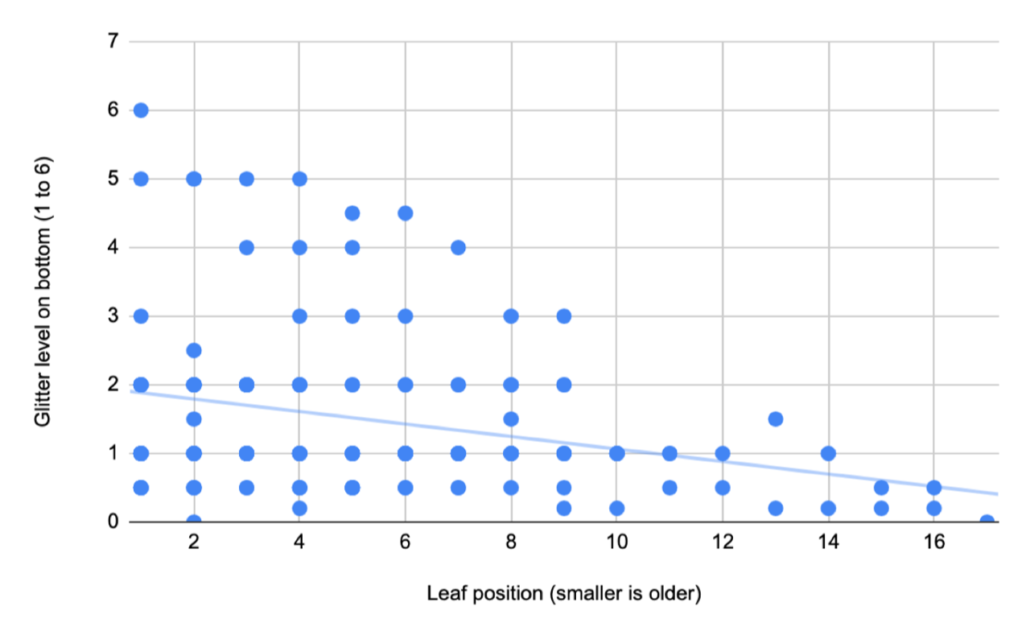
Loreli, a fourth time Young Scientist, chose a question based on her time at our school-year Nature Club. During one Nature Club session focused on nature journaling, Loreli made some close observations of the silvery scales on Autumn Olive leaves and wondered what the point of all that “glitter” was and why some leaves were “glitterier” than others. Based on background research, she came up with four hypotheses: 1. The glitter might help protect the leaves from sun damage. 2. The glitter might help protect the leaves from insect damage. 3. The glitter might help prevent the leaves from drying out. 4. The leaves might lose glitter as they age because it gets shaken off by wind and rain. After measuring the amount of glitter on many leaves and branches, Loreli found no relationship between sun, shade, insect damage, or glitteriness, debunking hypotheses 1. and 2. There was more glitter on the bottoms of leaves, where stomata are, so it may play a role in moisture retention, but surprisingly younger leaves actually had more glitter than older ones! As always, more research is needed!

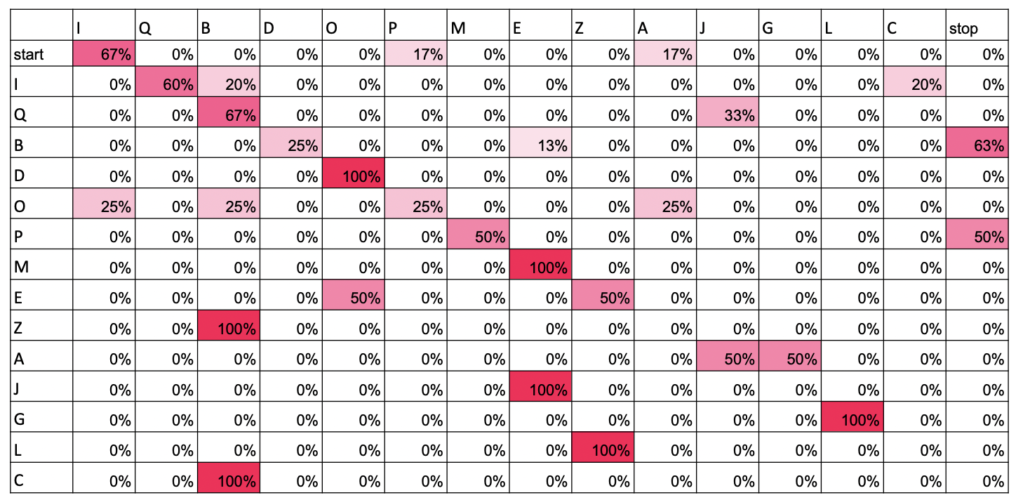
On the hike on the first morning, Luke, an avid birder, got interested in the songs of Red-eyed Vireos. Do they just repeat themselves over and over or do they mix up their songs every now and then? Do different individuals have different songs? When we actually set out to record vireos, we didn’t find as many as we were expecting, so we added Scarlet Tanager to the project, a serendipitous event. Luke made a “library” of phrases for each of the two species and used transition matrices to visualize how individual birds moved between phrases of their songs. Scarlet Tanagers tended to have very stereotypical orders in which they sang their phrases, while Red-eyed Vireos tended to be more random. Does this mean that the two species learn their songs differently? Or that females of the species have preferences for more or less variability? Other highlights of the week were adding Yellow-breasted Chat and Kentucky Warbler to Luke’s “life list.”

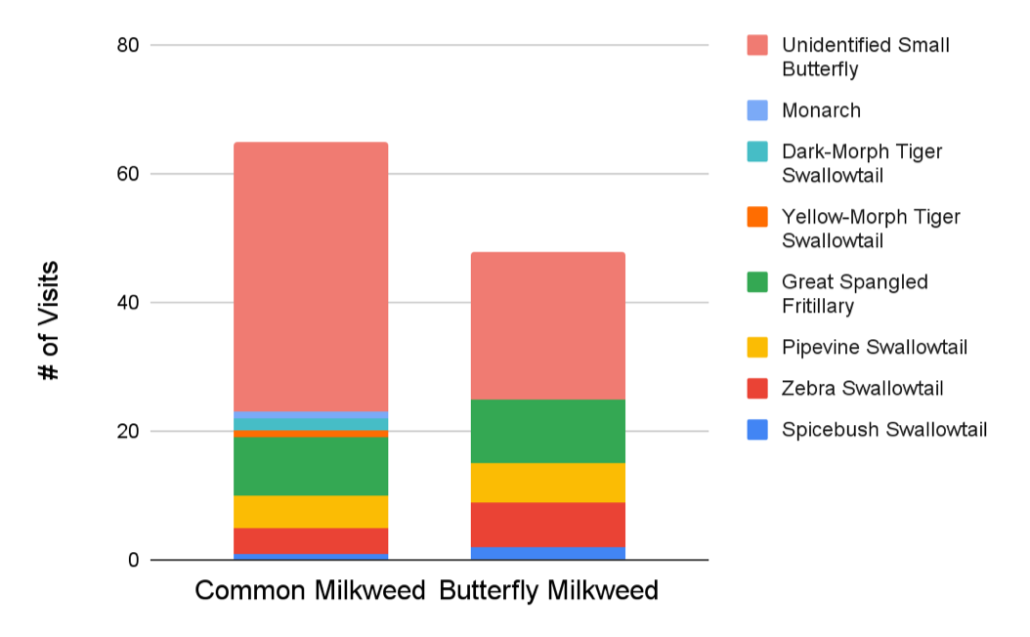
Myla got interested in all the butterflies flying around during a very hot week and wondered what attracted them to different types of flowers. One hypothesis was that they would prefer to hang out on dense patches with lots of flowers. Another is that they would prefer different colors. To test these hypotheses, she compared the butterflies visiting Common Milkweed (pink flowers in dense patches) and Butterfly-weed (orange flowers in sparser patches). Even after controlling for the number of flowers in a patch, butterflies in general seemed to have a preference for Common Milkweed, but there were more big butterflies on Butterfly-weed. Both species of milkweed are very popular, so pick your favorite color and plant it where you can!

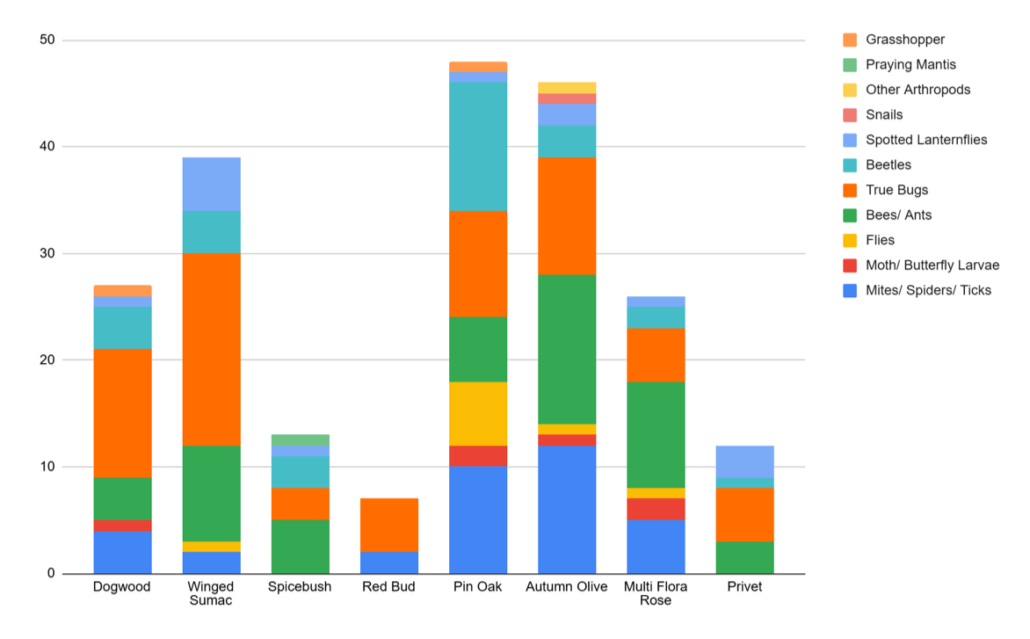
Nevin, a second time Young Scientist and Master Naturalist, is interested in conserving native plants and insects and wanted to study whether insects do indeed prefer native plants, as one would expect. He spent the week beat-sheeting different species of shrubs and counting the numbers of different orders of arthropods that fell off. Pin Oaks, a native species, did indeed have the most bugs, but surprisingly so did a few non-native species of plants. Nevin suspects that specialists insects, like caterpillars, may not have been captured very well with his study methods and that doing the same study at a different time of year would yield different results. One lesson learned was that there were a lot of ticks falling off of Redbuds so beware where you stand!

Insects are the most diverse group of organisms on earth, making up about 80% of all known species, but you don’t need to be an expert entomologist to appreciate these beautiful, wonderful, fascinating creatures that make up the foundation for every almost other form of life on earth. Here’s a quick guide to some of the most common insect orders. Just like with plants, it’s often easier to start by learning to identify a fly from a bee from a true bug (which can be harder than you think sometimes!) than trying to memorize individual species.
Download and/or print our free insect order guide here!
Every spring across the eastern United States a great migration takes place. But the creatures are small and they move under cover of darkness, so many people never see them at all. They are Spotted Salamanders and Jefferson Salamanders, who spend the winter in holes in burrows under the forest floor. Then, on the first warm rainy night of spring, they walk a few hundred yards to small pools of water where they will find each other, mate, lay eggs, and leave again before their babies hatch. Their jelly-like egg masses are tasty food for fish and other predators, so the salamanders rely on pools where fish and other aquatic animals can’t survive because they dry up in summer. These are called vernal pools or ephemeral pools.
Here at Clifton we have seven vernal pools. We have been making casual observations of our vernal pools for years, but in 2023 we started a long-term monitoring project to take quantitative data on our vernal pools and their residents over time. We want to answer the following questions:
1. Are our vernal pools changing over time and if so, how?
2. What kinds of vernal pools do amphibians prefer to lay their eggs in?
To answer those questions, we have enlisted the help of hundreds of children, college students, and adults to take data on the number of egg masses, water temperature, water pH, and water clarity as often as we can each spring.
This year we haven’t had a lot of rain, so the vernal pools are already drying up and the monitoring season is coming to a close. With the dry weather, some of our pools haven’t even had enough water for the amphibians to lay eggs. While three years of data may sound like a lot, one of the main lessons we have learned is that we will need several more years of data before we are able to say whether the year-to-year changes we are observing are part of a trend or just normal variation.
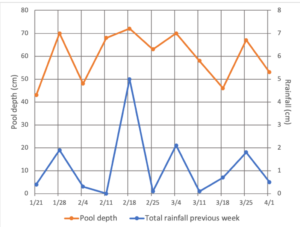
A more concrete lesson can be gleaned by comparing the depth of the vernal pools to rainfall data that has also been collected on the property. As shown in the graph, it takes about 2 centimeters of rainfall in a week to keep the pool we call Trifecta 1 full. Any less than that and the water level starts to drop. So it’s not just about total rainfall, but consistent rain throughout the spring.
If you or any students in your life would like to work through some other data from the project you can find a worksheet here.
Thank you to all the students and volunteers who have helped us collect these data! We are looking forward to seeing what we can learn in the next three years.




Words by Andrew Eberly
It’s been a few days since our first big snowstorm in a couple of years. We ended up with somewhere around 8 inches that arrived in two distinct waves of precipitation over the course of 24 hours. By now the original snowpack has melted, recrystallized, sublimed, compacted, and drifted in the wind so that we are left with an uneven 3 or so inches that still covers the entire landscape.
Despite the excitement of this latest storm, I would like to turn everyone’s attention to the brief but incredible blast of winter that occurred two days prior to the big storm. Friday, January 3rd began as a partly to mostly sunny day. It was cold early on, but the sun warmed up the ground throughout the day and by the afternoon ambient temperatures were well above freezing. I was out on the Big Hickory Trail scouting for the all-day hike the next day, sweating in my jacket, when the sky suddenly grew quite dark. Were we about to get a repeat of the highly anomalous severe thunderstorm event we saw on New Year’s Eve? Indeed, a sudden wind gust swept through as it might on a hot June afternoon in advance of summer storm. Instead of a warm rain, this gust front was bearing a burst of heavy snow. Soon the trees around me were obscured in the white-out and the ground was rapidly gathering a coating of little snow pellets called graupel. The graupel eventually gave way to a beautiful, fluffy, classic snow cascading out of the sky for about 30 minutes in which time we received a good inch and a half of accumulation.

This storm was what meteorologists refer to as a snow squall. That morning the NOAA forecast discussion stated that snow squall parameters were “off the charts” over the Allegheny Front in West Virginia. We now know that those parameters extended well east of there.
Like thunderstorms, snow squalls are a form of convection. Actually, if you were lucky enough to be in Prince William County on Friday, you were treated to one of the rare holy grails for weather enthusiasts, thundersnow! We just missed it here. Convection is the rapid and concentrated upward movement of air from near the surface to high in the atmosphere with subsequent downward movement nearby to balance things out. It indicates that there is a sharp difference in temperature between the relatively warm surface and cold upper atmosphere. This unstable situation can be caused either by moving a bunch of very cold air over top of warmer air or heating the air at the surface. Both things were occurring on Friday afternoon, so convection is what we got.
The type of cloud produced by convection is a cumulonimbus. These are tall clouds with frozen tops that are on the order of a few miles wide, a few miles high (much taller in the summer than winter), and they tend to occur in groups. Snowflakes form in convective clouds and then they get blown around inside the cloud by all the vertical air currents. As they swirl around, supercooled liquid droplets collide with the snow crystals and freeze onto the surface. Eventually they get covered in a frosty crust called rime ice. If you have climbed a tall mountain during the winter, you have probably seen objects covered in thick layers of rime ice from colliding with supercooled cloud droplets in the wind. When enough rime ice collects on a snowflake, it gets heavy and falls to the ground as a little pellet of graupel. Graupel is an indication of convective clouds. These same processes also occur in summer thunderstorms but the air at the bottom of the cloud is warm and melts the frozen precipitation.

Convection typically causes heavy precipitation, but it also doesn’t last long. After the Squall on Friday the sun came out and illuminated the freshly frosted landscape. This was immediately followed by another squall, then more sun. Such is the pattern that convective events tend to follow. Bursts of rising air with sinking air in between.
If you look at the temperature graph from the Clifton weather station for last Friday you can see that the temperature starts out quite cold, in the low 20s. After the sun comes up, the temperature climbs to a comfortable 42 degrees. The dewpoint also rises but not by as much indicating that the air is dry. Then around 3pm the temperature plummets but the dewpoint spikes briefly. This is the big snow squall. The dry air is what helped keep the snow from melting as it fell into 42-degree air. As water evaporates from the surface of the falling snow pellets or flakes, they cool down from the same process that cools you in the summer when you sweat. Eventually this cools the surrounding air and the temperature under the cloud drops. The creation of cold, dense air descending out of the cloud is also what causes the sudden wind gusts in convective events (indicated here by the even steeper drop in “feels like” temperature or wind chill). The spike in dewpoint is caused by the addition of water vapor to the atmosphere from the snow.

Habitat Specialist Andrew Eberly has many roles here, including growing seedlings for our plant sales, conducting plant and bird surveys, and helping to manage our 900-acre property. Here he shares some of the thinking that goes into taking care of our land.
Each fall I find myself out in the fields doing the repetitive, physical work of controlling the trees and shrubs that have worked their way into the 300 acres we are trying to manage as grasslands or shrubby savannas. This is time-consuming and sometimes arduous work, but it’s also enjoyable in many ways. It gives me a close look at what is growing out there and allows me time to ponder the pros and cons of the methods we are using to manage the land. What follows is a sample of some of these thoughts. To start with, there seem to be as many approaches to managing land as there are land managers and that’s OK. We are working with ecological processes that play out over tremendous periods of time and we must be at peace with the fact that a human lifetime isn’t long enough to see the results of all of our plans and hard work.

We’re all about grasslands here at Clifton. They are the most imperiled habitat we have and the plant species that comprise our remaining grasslands won’t survive if trees shade them out. The easiest thing would be to mow them short every year, but transitional habitats are also valuable. It’s a constant struggle to strike a balance between what is feasible from a management perspective and creating a mosaic of multiple successional stages.
The field we call Turkey Gap is an old pasture in the process of diversifying and reverting to a wilder type of shrubby savanna ecosystem. It’s a good example of the kind of transitional habitat that we want to make sure persists. Most of Turkey Gap gets bush hogged regularly, but for the last three years I’ve set aside a small area to see if there’s any chance of maintaining it without mowing. Mowing is a necessary tool in grassland management, but in my experience it has some downsides. For small animals living in fields, mowing is a catastrophic event. Also some invasive woody plants actually increase when they get mowed once a year. Finally, mowing has a tendency to smooth out the natural heterogeneity of sizes and ages naturally found in grasslands and shrublands.
My experiment now hosts hundreds of Tulip Poplars, Black Walnuts, Autumn Olive, and many more tree species that have already reached 15’ to 20’ in height. The result is a beautiful early successional woodland that hosts nesting American Woodcocks, roosting Red Bats, Box Turtles, and countless other animal species that seem to depend on having some scraggly, immature woodland patches within their home ranges. But soon my little section of Turkey Gap will grow up into mature forest unless I do something about it. Removing tree seedlings by hand with loppers and spraying the stumps with herbicide is the most targeted approach. Moving slowly through the habitat in this way allows me to find and avoid killing special native shrubs like Southern Crabapple and American Plum, along with more common species like sumacs and native roses that I believe should have a place in any savanna. Basal bark spraying with herbicide is a quicker strategy I plan on exploring in the future.

Why do I have to use chemicals anyway? Most of our perennial broad-leaved plants are adapted to being broken, burned, or cut periodically and they will happily resprout repeatedly with more individual stems each time. Invasive Autumn Olive is especially good at the “hydra” growth pattern. Herbicide is necessary to prevent the trees I’m removing from growing right back. Ultimately we are trying to mimic the natural disturbances of fire and grazing, so why don’t we just burn it and put some Bison out there? We will! Well, maybe not bison. Bison are a challenge to contain and they eat mostly grass, so they are hard to work with. We do prescribed burns, but they can be logistically hard to pull off. Fires are particularly challenging during the growing season when they would have the biggest impact on tree growth. Plus, in many parts of the Clifton property–even places that get burned repeatedly–woody species sometimes keep growing and eventually dominate to the point where they inhibit the growth of more flammable grasses and wildflowers, which prevents fire from having the effects we want it to. Some spots simply don’t want to burn.
It seems that if we want grasslands and savannas alongside forests on a medium-sized property like ours we have to be a little heavy-handed. I’m all for “letting nature take its course” in large wilderness areas where there will naturally be sections at different stages of succession, but we have to work hard to create and maintain the diversity of habitats we want here.
This kind of thinking leads me to another question that I often think about when I’m out there. How feasible is it to maintain native grasslands here long-term? The Clifton Institute lies on metabasalt (greenstone) bedrock. The soil here is relatively nutrient rich and some spots hold water for a long time. There aren’t as many remnant prairies on metabasalt compared to the poorer soils to our east, apparently because most sites have been converted to agriculture. These richer and moister soils tend to benefit non-native plants, which makes my job harder.
On the Clifton Institute property, we are living with the legacy of hundreds of years of agriculture. The current placement of our fields and forests is dictated by this history, as is the species composition of our plant communities. Moving toward a diverse mosaic of habitats dominated by native species will take time. We will continue to use the tools at our disposal and take cues from the environment to guide things toward greater biological diversity. This is a process that will take many years and will hopefully extend far beyond any of our lifetimes.

WARRENTON, Va.—On October 19, 2024, the Clifton Institute will host a grand opening and bioblitz for an exciting new project, Lawn Lab. In partnership with Fauquier County Public Schools and the Bee City USA committee, the Clifton Institute is expanding its grassland research program by setting up a land management experiment on the campus of Grace Miller Elementary School and Liberty High School. Through Lawn Lab, students at these schools will get hands-on experience doing environmental research in their own schoolyard. During the bioblitz, participants will record the species of plants and animals present in the study plots, collecting the first year of data for what will hopefully be a multi-year project. The event will begin at 2 p.m. and close at 4 p.m., with a brief ceremony, remarks, and refreshments scheduled for 3:30. This event is free and open to the public. Please park at Grace Miller Elementary School.
The Clifton Institute hosts field trips throughout the year where students learn how our land management choices affect native plants and animals. Some land management methods being researched are not applicable to every property (for example, prescribed burns,) However, lawns are ubiquitous and something that many landowners can choose how to manage. Some research has found that reducing mowing can have a positive impact on native plant and insect populations, but the students participating in Lawn Lab will help collect much-needed data.
“We love having students out to our property for field trips,” says Co-Director Eleanor Harris,“but they usually only get to come here once a year. With Lawn Lab, they’ll be able to go out and explore a wildflower meadow all year long and they’ll be doing real research on how their own schoolyard gets managed.”
The campus shared by Liberty High School and Grace Miller Elementary School in Bealeton includes a disused three-acre playing field. Two acres have been set aside to be mowed only once a year. The third acre will get mowed weekly as usual. Students will study these experimental plots throughout the year. This project is a rare example of a win-win-win-win: students will spend time outside doing field work just like staff scientists at Clifton, the school campus will become better habitat for pollinators, the county will spend less money and cut down on pollution by mowing less often, and the experiment will serve as a demonstration plot for the community and future projects. This initiative also supports Fauquier County’s mandate as a Bee City USA to create pollinator habitat.
###
The Clifton Institute is a 501(c)(3) non-profit organization in Warrenton, VA. Our mission is to inspire a deeper understanding and appreciation of nature, to study the ecology of our region, to restore habitat, and to conserve native biodiversity. We provide environmental education, conduct ecological research, and restore habitat for native plants and animals. The institute’s 900-acre field station, which includes forests, grasslands, shrub fields, and wetlands, provides a beautiful and easily accessible location for education programs and is permanently protected under a conservation easement.
Words and photos by Bert Harris
There are more than 400 species of lichen found in our area. They are varied and beautiful, and they serve as important habitat for myriad creatures. But they don’t receive much attention from naturalists compared to other groups. The lichens of the Blue Ridge to our west and the Delmarva Peninsula to our east have received recent study, but we still have a lot to learn about lichen communities in the Virginia Piedmont.
In September, we co-hosted the 33rd Tuckerman Lichen Workshop with James Lendemer from the New York State Museum. The goals of the workshop were to study lichens in northern Virginia and to help amateur lichenologists and graduate students hone their skills. The workshop is named after Edward Tuckerman, the father of North American lichenology.

The Oak Spring Garden Foundation (OSGF) generously put up 14 lichenologists and lent microscopes and space for them to set up a lichen identification lab. The group spent five days exploring sites with varied geologies and habitats: the Bull Run Mountains, Riverside Preserve, Wildcat Mountain, Calmes Neck, and the OSGF. The group braved wet conditions and a highlight was a rainy ride in the back of a pickup truck to access some distant lichens.

We are still identifying specimens but it seems that we found at least two undescribed species(!) and a handful of new state records. Other highlights included quartzite specialists in the Bull Run Mountains, three species of cyanolichens at Wildcat Mountain and Riverside Preserve, rarities such as Heterodermia granulifera in the forest at Oak Spring, and a diverse community limestone specialists at Calmes Neck. Overall, the experts were pleasantly surprised by how many species we found. Check out this link for photos from the workshop.
The workshop was part of a broader National Science Foundation award to James Lendemer to study Appalachian lichens. We are so grateful to all the lichenologists who attended, from as far away as Ottawa and Seattle. We also extend our thanks to the Oak Spring Garden Foundation for being such generous hosts. Many thanks also to private landowners, the Virginia Outdoors Foundation, the Department of Conservation and Recreation, and the Fauquier County Department of Parks and Recreation for allowing property access.

We weren’t sure what to expect as we headed into our second year of seed collection for the Virginia Native Seed Pilot Project. Finding suitable wild populations for each ecotype we produce (coastal, mountain, piedmont) is difficult to begin with, and we wanted to give last year’s collection sites a break this year to make sure there are no negative impacts on the wild populations. This was the last full year to collect the material needed for the current scope of this project, so it was imperative that we used every resource possible to collect all 18 of our target species. Luckily, some key collaborations helped save the season.
A huge part of this year’s success was due to the Virginia Master Naturalists, who established a statewide project to help us with seed collection. VMN volunteers collected more than a third of our entire final seed supply. We also had assistance from several public schools and other independent volunteers who made time to collect seeds for the project!
Additionally, the Department of Wildlife Resources gave us permission to collect seeds at over a dozen different wildlife management areas. With the dedication of our many volunteers, help from VMN, and the partnership with DWR, we were able to collect all 18 target species and we expanded our reach into mountain and coastal areas.

As we wait for spring and the next growing season, we are working on securing additional funding to extend the project another three years. This is the critical next step to see more Virginia ecotype seed hit the market because it will help us get our harvesting operations to the commercial scale.
We’re planning to expand the project by recruiting more farmers in the western portion of Virginia, responding to interest from several public schools regarding implementing programs for native plant propagation, and collaborating with the Rappahannock Tribe to establish a seed harvesting operation. All of this interest helps reaffirm what we know about this project–it has a broad range of applications and there are countless ways that it can benefit everyone from the farmers who grow the seeds, the consumers who plant them, and of course, the wildlife that depend on native plants.

Thank you to our partners and collaborators thus far:
The Nature Conservancy
Ernst Conservation Seeds
Capital Region Land Conservancy
Department of Conservation Recreation
Virginia State University
Department of Wildlife Resources
Center for Urban Habitats
Virginia Master Naturalists
Friends of Dragon Run
Chancellor’s Rock Farm
Slade Farms
Pat Acres Farm
Morning Glory Farm
Rev. Dr. Johnny & Fonda Hicks
Marky Dewhirst & Scott Barboza

